Tilapia farming
Introduction
Aquaculture or more specifically tilapia farming comes in all sizes from large commercial producers to small backyard ponds. While they all share a few common ingredients, obviously water and tilapia, the equipment and methods used are different for each. It is unlikely for example that you would find an oxygen generator, cyclone filter, drum filter or an ion exchange and electrochemical regeneration system for removing ammonia on a back yard tilapia farm. Conversely, you probably wouldn't find any air stones, filter pads or bio balls in use at a commercial aquaculture facility. An important concept for you to keep in mind as you make your way through this guide is: that no single method described is better than the other when it comes to your own farm. Just like shoe sizes, there is only one exact fit and everything else is either too big or too small.
In recent years there have been a few manufacturers who have developed "expandable" aquaculture and aquaponic systems for commercial use, but these are just small systems using small farming methods set up in repetition. These systems are not an economically viable alternative to purpose-built facilities designed to exactly meet the desired production output from the onset. Additionally, there is a wide variety of computerized controllers and testing equipment that was originally developed for industrial sized aquaculture and is now being marketed to backyard tilapia farmers. In smaller systems with relatively low volumes of water and rapidly changing chemistry, using pricey testing equipment may not deliver the expected benefits. This may result in tilapia farmers who continually chase down inexistent water "problems" and produce filets that cost a fortune.
We created this guide for anyone interested in tilapia farming. Wherever we describe a process, we will include methods for both large commercial production and backyard tilapia ponds. We will use bullet points, the little red fish, to reiterate points that we think are important for you to remember. We'll also use green boxed text to add additional comments that are either critical or applicable to aquaponic growers. This guide will evolve as new methods are researched and then published in the Journal of Applied Aquaculture or other reputable publications. We invite you to contact us about anything that we haven't made perfectly clear so that we can update this guide for the benefit of everyone. We wish you success.
Raising tilapia
From the tilapia farmer's perspective, there are three main events in the tilapia farming timeline: hatching, rearing and harvesting. Of course, these events have many different names depending on with whom you are speaking. Some people might use words like spawning, grow-out and processing, but no matter what terms they use they're all talking about the same things. An important point to remember is that we are referring to the events and jobs in the farming timeline and not the development cycle of the tilapia. Although they are interwoven, the tilapia are going through their own cycle of development that doesn't require any significant shifts in your responsibilities. Because this is an important distinction we will briefly overview each of the tilapia farming events.
- Hatching includes delicate jobs such as caring for breeding colonies, encouraging or inducing spawning, egg extraction or nursery isolation, tilapia fry care and raising the fry to fingerling size. Ultimately grading the fingerlings for their rate of growth before delivering them to the grow-out facility. Each of these jobs has several individual steps and techniques that are unique to the operation of a tilapia hatchery. It should also be noted that the equipment and facilities used for hatching are unique to hatchery operations and only useful during the first few weeks of the tilapia’s life.
- Rearing or grow-out is the part of tilapia farming that picks up after the hatchery has raised them to fingerling size. At this stage the tilapia farmer's goal is to raise the tilapia to harvest size quickly, economically and in good health. Tasks include testing, sorting, weighing and several maintenance jobs. These tasks are the subject of this guide.
- Harvesting or processing involves selecting tilapia, moving them to a finishing pond, killing them humanely in a way that respects what they are providing and then removing their filets. Many of these jobs can be skipped by the farmer and passed on to the person preparing the tilapia. The equipment used for harvesting has nothing to do with the rearing facilities and obviously doesn’t incorporate any of the hatchery equipment.
So as you can see, hatching, rearing and harvesting not only involve completely different sets of responsibilities, they also require different equipment and facilities. It should also be noted that the size of the operation doesn’t matter. For example, a processing facility can be as complex as climate controlled clean room full of stainless steel tables and equipment or as simple as a home kitchen with a sink and a cutting board. Every tilapia needs the same things to live and the only difference between the large commercial farm and the backyard farm, are the methods used. In the end, results are all that matter. The level of creativity that you use to get there is up to you and part of the personal satisfaction that you'll get from tilapia farming.
What follows is intended to be a kind of need-to-know, answers-only guide to tilapia farming. We're not going to fill your head with theory and science beyond what is absolutely necessary. In addition, we are going presume that you have an average level of common sense. With respect to book writers, who have to fill pages with text by first stating and then repeating the obvious, sentences such as "the tilapia go into the pond" are not a part of this guide. So without further ado, let's learn about tilapia farming.
Point of clarification: Throughout this guide we use the word "pond" to describe a container of any size that holds tilapia.
The five needs of tilapia
Tilapia don't ask for much. In fact they only have five basic needs: clean water, oxygen, food, light and room to swim. Give your tilapia these things and they will stay healthy and grow fast. The art of tilapia farming is to understand each of these needs and then find a way to provide them in sufficient quantities. The problem is that each of these five needs comes with a myriad of potentially complicated questions and solutions. In the next five sections we will address each of the tilapia's needs one at a time.
Aquaponics Point: Tilapia do not care what you do with their poop or how you remedy ammonia and nitrate contaminated water. It does not matter whether your operation is straight aquaculture or if you use your tilapia's pond water to grow plants. Aquaponics is not a new way to raise tilapia; it is an alternative way to deal with and potentially benefit from fish waste. Of course, if you were to ask vegetable farmers they might tell you that aquaponics is a novel way to fertilize their plants. But regardless of your perspective, in all farming situations the needs of the tilapia always remain the same.
Tilapia need number one - Clean water
Providing your tilapia with clean water can be split into two parts: new water introduction and existing water maintenance.
New Water Introduction
Whenever you introduce new water into your pond or aquarium, it needs to be of the same quality that you would drink yourself. In fact, if you aren’t willing to drink the water that you are introducing to your tilapia, then you need to stop giving it to them until you are. Tilapia are a food fish, so whatever is in their water, will eventually wind up in your body. You might as well drink the water now and cut out the middle-fish.
Incredibly important point: Tilapia do not drink water. Like all freshwater fish they absorb water through their skin and gills by osmosis. Whatever is in their water will be absorbed into their bodies.
Your water should only come from a safe municipal source or a clean private well. If you only buy bottled water because you can’t stand the taste of your own local water then do something about it. Buy a filter, a softener, a nitrate remover or a high volume reverse osmosis system and do whatever it takes to get the water to a condition that you would drink yourself.
Critical Point: Never ever use 100% reverse osmosis water for tilapia farming purposes. Aside of the fact that RO water will destroy some testing equipment, like pH meter probes, it has no buffers for pH fluctuations. A carbonate hardness of between 150 and 350 ppm is necessary for tilapia health.
After you are happy with the drinkability of your water, fill up a food safe transfer container or tank to further treat the water before you give it to your tilapia. It's a bad practice to simply run hoses from the water source directly to your pond. Sudden changes in temperature, pH or other water chemistry originating at the source is common. This can stress tilapia, causing weakened immune systems and could even upset the balance of established biological colonies. The size of the transfer container is up to you, but we recommend that it be able to hold at least 20 percent of the volume of your pond. For commercial operations, 100 percent is recommended.
As you are filling your transfer container(s), you need to make sure that the water you are going to add to your tilapia pond is at the same temperature as the water to which your tilapia are already accustomed. Plus or minus a couple of degrees is okay, but if the difference is too great it will shock them.
- Make sure that you are willing to drink from your water source before you give it to your tilapia.
- Put new water into a transfer container for further treatment before adding it to your pond.
- Make sure that the temperature is the same as to what your tilapia are already accustomed.
In addition to making sure that the newly introduced water is clean enough for you to drink and at the right temperature, you'll need to make sure that the water is free of all chemicals added by the municipal water authority, especially chlorine or chloramine. A gallon jug of Aquatisafe™ aquaculture pre-treatment goes a long way when you consider that it treats 35,000 gallons and removes chlorine, chloramine and reduces the toxicity of heavy metals like copper, cadmium, mercury, silver, zinc, lead, nickel, manganese and sodium selenate which can all be present in any water supply. Also, do not assume that treated municipal water will lose its chlorine content on its own over time. This is especially true for water treated with chloramine. Even if you can't smell the fumes it only takes trace amounts to cause deadly chemical burns to their gills and throughout their bodies.
- Use a food fish safe water pre-treatment such as Aquatisafe™ to remove chlorine or chloramine and reduce heavy metals in new water before it is introduced into your tilapia pond.
- Do not rely on time or aeration to remove chlorine from your water.
Note: Aquatisafe™ is a professional product used for food fish production. Consumer equivalents can be found in pet stores that sell aquarium supplies and is generally sold as "tap water conditioner". Just make sure that the label reads that it is safe for food fish. If you want the brand that we use, contact us and we'll be happy to help.
You also need to make sure that newly introduced water is at the ideal pH level and that it is at the same pH level of the water already in your pond. This may seem like a strange way of saying it, but the wording is intentional. Fish keepers tend to get into a bad habit of adjusting the pH level of their ponds to ideal by introducing new water with a significantly higher or lower pH. Their hope is that when the new water is added to the old water the differing pH levels will mix and result in the target pH. This is the equivalent of throwing phosphoric acid and potassium carbonate at someone in the hopes that the two will cancel each other out and achieve some sort of perfect balance.
Important point: Phosphoric acid and Citric acid should be used to lower pH and Potassium carbonate should be used to raise pH. Many acids and bases are dangerous to fish and humans. Always use food grade acids and bases.
The proper procedure is to test the pH level of the water in your pond and use pH-Down or pH-Up to bring the existing water to the ideal level slowly. At the same time adjust the water in your transfer container(s) to the same ideal pH level. Be sure to read the labels of all of the products or chemicals that you want to use to make sure that they do not read "not intended for food fish" on the warning label. Once the new and existing waters are at exactly the same pH (and temperature) level you can move on to the next treatment step or safely drain off the existing water and introduce the new water to your tilapia.
So the obvious question is: What is the ideal pH level for tilapia? The easy answer is 8.0, but there are some common situations that make 8.0 impossible. Many plants in an aquaponic system prefer a pH closer to 6.0 and since the fish and plants share the same water a pH level of 6 or 7 (point) something becomes the ideal. We’ve seen some ponds that due to their construction and alkalinity, rapidly creep to about 8.4 and stay there no matter how many times the water is treated back down to 8.0. In those cases we stop fighting the losing battle and just make 8.4 the new ideal. It is far better to let the fish swim in a pH of 8.4 than it is to constantly hit them with pH changes.
Critical Point: The extreme pH ranges for tilapia are between 3.7 and 11 and the pH ranges for optimal growth are between 7 and 9. However, a more toxic form of ammonia, known as un-ionized ammonia (NH3), is produced in water with a higher pH (and temperature) level. The other variety, ionized ammonium (NH4+), is not toxic. The pH of water changes with alkalinity and also fluctuates with carbon dioxide levels which rise and fall with photosyntheses. We therefore recommend that you keep your pond between 6.5 and 8.0 to mitigate potential losses due to a spike in ammonia. Also, because pH and Ammonia are cyclic, we recommend that you only test pH and Ammonia in the late afternoon.
- Adjust the pH of the water in your transfer container and your pond to ideal before you introduce the new water to your tilapia.
Finally, you should match the salinity of newly introduced water to the existing pond water. Although not necessary until problems arise, many tilapia farmers add a small amount of non-iodized salt (NaCl) to their water to aid in the prevention of parasites and to mitigate the problems associated with elevated nitrites (Brown Blood disease). Adding salt to a measurement of 6 parts per thousand or to a specific gravity of 1.004, which is roughly one tablespoon of salt per gallon of water, will prevent most parasites from developing. Of course, for every level of salinity there are parasites that can thrive, but the purely fresh water parasites seem to develop the earliest.
Critical Point: Never add salt in any amount to a system that utilizes Clinoptilolite or any other zeolite for ammonia removal. Doing so will cause the absorbed ammonia to be released back into the water.
While we're on the subject of salt, some old-timers might tell you that sodium bicarbonate (NaHCO3) or epsom salt (Magnesium sulfate, MgSO4) can or should be used instead of common table salt (NaCl), but this is incorrect. Sodium bicarbonate is used as a temporary buffer for fish hauling and shipping purposes and epsom salt has limited uses in aquaponic systems and is of no use to fish farming operations.
Critical Point: You can safely add salt (NaCl) up to 36 parts per thousand for Blue and Mozambique tilapia however the recommended maximum for optimal growth is 19 parts per thousand. Nile tilapia are not as tolerant to saline water. Nile tilapia should not be put in water containing salt levels above 18 parts per thousand.
- If necessary, match the salinity of any new water to your pond before giving it to your tilapia.
Aquaponics Point: While aquaponics can significantly decrease the frequency of traditional water changes or eliminate them entirely, the action of adding water lost to evapotranspiration (look it up) is effectively a water change in itself. Fresh, clean water contains many trace minerals that are beneficial to both the tilapia and to the plants. Use a good nitrate test kit periodically, just to be sure that your plants are keeping up with your fish. Also, since we mentioned epsom salt above, never add more than three parts per thousand of epsom salt to your aquaponic system.
Existing Water Maintenance
The water that your tilapia are swimming in will never be cleaner than when you first introduce it into their pond. From that point forward your pond water will continue to get more and more toxic, until it kills your tilapia, unless you intervene by removing the old dirty water and introducing new clean water into their pond. Most people are surprised to learn that many fish farms, particularly trout and salmon farms, use no filtration or treatment whatsoever and instead rely on constant water changes. This is normally accomplished by diverting water from a nearby river through the fish ponds and back out again in a continual flow. Another method is to do away with the pond altogether and just raise fish in large suspended nets out in the middle of a lake or slow moving river. In fact, you can even raise tilapia in an aquarium at home without any filtration or treatment at all provided that you are willing to replace their water every single day. But honestly, who has that much free time?
- Tilapia do not need filtration to thrive as long as you are willing to replace their water every day.
For those of us who don’t want to do daily water changes, there are ways to delay the task for days, weeks or even months by using filtration and treatment. In fact, the only purpose of filtration and treatment is to buy yourself some time between water changes. How much time you get depends entirely on how efficient the filtration or how effective the treatment is. For the rest of this section we will go over some of the common things that make tilapia pond water toxic and what you can do to delay or prevent their build up, so that you can reduce the frequency of water changes.
- Filtration and treatment are used to convert or reduce toxic compounds in aquaculture water, thereby reducing the frequency of water changes.
Undissolved solids are the first things that will begin to make your tilapia pond water toxic. This is the stuff that you can easily see suspended in the water or resting on the bottom. Basically, it’s uneaten food and tilapia poop. These solids will eventually dissolve into the water becoming dissolved solids and will contribute to the build up of toxic compounds such as un-ionized ammonia. The best way to trap these undissolved solids in a small to medium sized system is to use a pre-filter that passes the water through a barrier material. Disposable or serviceable (cleanable) filter pads or screens are typically used for this purpose. For large systems, a drum filter will be necessary. If applied correctly these methods should capture nearly all of the undissolved solids in your system before they become a problem.
It's important to emphasize, hence this red text, that pre-filters do not remove solids, they only trap them. Until the solids are actually removed either manually or by a drum filter they will continue to dissolve and contribute to the toxicity of your pond water. There is no contraption that will remove tilapia poop other than a drum filter. You must clean every other type of filter by hand.
- Undissolved solids are only trapped by pre-filters, they are not removed.
- Undissolved solids that are left in pre-filters contribute to pond toxicity (un-ionized ammonia) until they are removed.
Dissolved solids are comprised of food and poop which has been broken down into very fine particles that remain suspended in the water and pass through pre-filters. Dissolved solids contribute to the formation of other more toxic compounds such as un-ionized ammonia. The best way to trap dissolved solids for most aquaculture is with the use of a fine-particle barrier filter. On very large fish farms, where the volume of water is closer to that of a small city, chemical processes may be used to remove dissolved solids as part of a separate water treatment and reclamation system. Like pre-filters, fine-particle barrier filters do not remove dissolved solids by themselves. You must service these filters to remove the contaminants. We will refer to this filtration step as "fine-particle" throughout this guide.
There are other dissolved contaminants such as tannins and phenols which can color your pond water and give it a smell. These contaminants are caused by organic matter and are so small that they pass right through even fine-particle barrier filters with ease. Think of how a teabag affects water when you make a cup of tea. The only way to remove these microscopic particles is with activated carbon or with the chemical treatment processes only used on the largest farms. Unfortunately activated carbon is exhausted very quickly and can be relatively expensive to replace so it's not practical for constant use. Our opinion is that activated carbon should be only used on an as-needed basis on smaller tilapia farming operations to clarify tea colored water or reduce odors. Activated carbon is not an economically viable solution for commercial tilapia farming use.
- Fine-particle filters only trap dissolved solids they do not remove them. You must clean your filter media to stop the dissolved solids from making your pond toxic (with ammonia).
- Activated carbon is useless against dissolved solids, but can be used to trap tannins and phenols in smaller ponds.
Un-Ionized Ammonia is the first truly deadly compound that you will encounter. Un-ionized ammonia is produced by decomposing organic matter and healthy tilapia in water with a pH above 7.0. The only way to remove un-ionized ammonia is to replace the water or find a way to eliminate the ammonia. The good news is, there are naturally occurring bacteria that readily consume ammonia. The bad news is, the ammonia-eating bacteria (Nitrosomonas) give off even deadlier compounds, called nitrites. Nitrites oxidize hemoglobin into methemoglobin making it difficult for your tilapia’s blood to carry oxygen (hypoxia) and will cause suffocation at the slightest exertion. Fortunately for the tilapia the nitrites are further oxidized into something far less lethal, called nitrates. Once nitrites have been converted into nitrates, your tilapia are out of immediate danger. Over time, however, the nitrates will build up in your pond and you will finally have to do the dreaded water change.
Critical Point: Test kits and equipment read the "total ammonia" but this has nothing to do with the level of toxic (NH3) ammonia present in the water. The level of toxic ammonia must be calculated in conjunction with the pH level and temperature. At room temperature with a pH of 6.0, all of the ammonia is basically non-toxic. At a pH of 8.0, only about 10 percent or less is toxic. In fact, you have to raise your pH to 9.0 before the total ammonia is only half-toxic. What's the hidden lesson in all this? You can control the toxicity of ammonia using pH!
- For the rest of this guide the reader should consider all references to ammonia to mean the toxic un-ionized ammonia unless otherwise specified.
Another Critical Point: Ammonia is toxic to Blue tilapia at concentrations above 2.5 milligrams per liter and above 7.1 mg/L for Nile tilapia. However, ammonia concentrations as low as 0.1 mg/L will depress food intake and growth. Always strive to remove toxic ammonia completely from your system. Even small amounts can cost money in the form of longer grow out periods and wasted food.
The nitrifying bacterium, called Nitrosomonas, responsible for oxidizing ammonia into nitrite and a bacterium called Nitrobacter, which further oxidizes the nitrite into nitrate, live on every surface of your pond along with many other types of bacteria. Some of these bacteria are aerobic, meaning that they need oxygen and some are anaerobic, which means that they grow in conditions with very little oxygen. Normally, you will find the nitrifying bacteria along the water line in your pond as well as every under water surface and inside pipes. Unfortunately, that's not nearly enough surface area to support the number of bacteria colonies needed to convert the amount of ammonia being produced. The solution is a contraption commonly referred to as a bio filter or bio reactor.
Bio filters have only one purpose: to give a whole lot of surface area for nitrifying bacteria to grow on. The two most popular bio filter medias are bio sponges and bio balls. Other good bio medias include stranded PVC and bio straws. Unlike the filters designed to trap undissolved and dissolved solids, the bio media should not be serviced until the water flowing through is being restricted. Even then, they should just need a light rinsing to get the water passing through them again.
Aquaponics Point: Your grow bed media is your bio filter unless you are only using floating rafts. In aquaponic systems that only use floating rafts, we recommend that you incorporate a bio filter somewhere in your plumbing. For example, after your solids separator or between your sump and fish tank. Your grow bed media should be designed to prevent conditions for anaerobic bacteria growth as these conditions are also detrimental to plants. In other words, ensure good water flow and avoid stagnate pockets of water.
- The bio filter only provides a growing surface area for the nitrifying bacteria that eliminate toxic ammonia.
- Never clean or sanitize your bio filter, just rinse it lightly if it is restricting the flow of water.
Important point: There are certain tilapia farming situations where it is not practical or even possible to remove ammonia using bacteria. Aquaculture tanks or ponds that are not part of an aquaponic system can be successfully farmed using alternative ammonia elimination methods such as zeolite or aggressive aeration. By removing ammonia, the need for ammonia-consuming bacteria is also eliminated. These methods also prevent nitrites and nitrates from being created, so water changes are no longer required for nitrate removal.
The final step in providing your tilapia with clean water, has to do with the prevention of parasites and pathogens. If you don’t take measures to prevent them, parasites might happen to your tilapia at some point. As we mentioned earlier on this page, if you get caught with parasites, you can kill them pretty easily, without hurting your tilapia or ruining their food value, by changing the salinity of the water to 6 parts per thousand, using non-iodized salt. This will wipe out the parasites very quickly. It should also be mentioned that, if you are raising your tilapia in water that already contains salt and you get a parasitic outbreak, you can put your tilapia in fresh water to kill the parasites. In a nutshell, parasites can’t handle sudden changes in salinity.
If your tilapia get a pathogen (disease) however, it’s game over. Euthanize your tilapia because they are going to die anyway. Then drain your pond, disassemble your filtration and sanitize the expensive parts with an acid-based sanitizer, throwing everything else away. No, we’re not kidding. It is illegal in the United States to sell a food fish that has been treated for any disease and for good reasons. Many pathogens are untreatable and those that are treatable, require expensive injections, that cost more than the tilapia themselves and must be administered individually. Not to mention the fact that the incubation period for most pathogens, is longer than it takes for the tilapia to grow to harvest size. So it might not be clear by looking at the fillets if they still had the disease at the time of harvesting and processing. Pathogens are all-around bad news in tilapia farming.
Critical point: The length of time that a tilapia can survive with a pathogen is directly related to it's age, size and immune system. Tilapia fry and fingerlings weighing one gram or less have no resistance to pathogens whatsoever and will die almost immediately after exposure, whereas larger tilapia can survive for much longer. This is why testing for pathogens in harvest size tilapia is so important and also why testing for diseases in tilapia fry is completely useless. The simple fact that the tilapia fry are alive is evidence that they have no disease.
For our purposes, we've included viruses in with pathogens to keep the discussion simple even though they infect tilapia in different ways. When is comes to disease, it is far more practical to concentrate your efforts on prevention, rather than reacting to an outbreak. The first step in prevention, is to reduce the risk of getting them in the first place. The following is a list of the preventative measures that we suggest:
- Sanitize your hands and arms before putting them into your pond water.
- Use gloves.
- Maintain clean conditions around ponds. Sanitize the floors of indoor areas and sanitize the bottoms of shoes if practical.
- Keep separate sets of equipment, such as nets and buckets, for each pond.
- Adopt a colored bucket system. White for clean water and fish holding, blue for equipment and filter cleaning or carrying and gray for toxic water carrying.
- Avoid conditions that cause weakened immune systems in tilapia, such as stress due to overcrowding, poor nutrition and high levels of nitrates.
- Prevent pets and other animals, from drinking from your tilapia pond water.
- Keep birds from pooping in your tilapia pond.
- Do not put snails, shrimp, goldfish or any other living organisms, in your tilapia pond water.
Critical Point: Never ever put tilapia in a system that is occupied by snails or goldfish. Snails and goldfish carry parasites that are foreign to tilapia and will kill them. You may get away with it a few times, but eventually the odds will catch up with you. Once these parasites have found refuge in your system, it will need to be completely sanitized to remove them.
An ultraviolet sterilizer is the single best piece of equipment that you can use to control parasites and pathogens in your water, before they can get into your tilapia. By passing water in close proximity to an ultraviolet light source, a properly sized UV sterilizer kills just about everything. The key to successfully sterilizing your pond, is to expose the right volume of water to the UV light source for the correct amount of time. In the case of ultraviolet sterilizers, larger and higher wattage does not necessarily mean better. It is important to select one that is the correct size for your pond and then make sure that you adjust your plumbing to the manufacturers recommended water flow rate. You can time how long it takes for the water coming out of your UV sterilizer to fill a five gallon bucket, to determine the flow rate and then adjust it with a ball valve in front of the inlet if necessary.
- Parasites can be eliminated from your tilapia and your water with salinity changes.
- An ultraviolet sterilizer can only remove parasites and pathogens from water, not from the tilapia themselves.
- Eliminating parasites and pathogens in water will prevent them from transferring between individual tilapia.
- Individual tilapia with parasites can be treated by placing them in a tank of water containing 6 ppt of non-iodized salt for a few hours.
- Pathogens in tilapia cannot be treated effectively, economically and in some cases legally. The only viable answer is prevention.
There are a couple more things that are worth mentioning about ultraviolet sterilizers. First, they are the only realistic option for preventing parasites in aquaponic systems. Some aquaponic dealers pretend that parasites and diseases don’t happen, but this has more to do with salesmanship than anything else. After all, a car salesman doesn’t show pictures of people injured in car accidents as part of his advertising, so it’s understandable. But tilapia growing in aquaponic systems do occasionally get affected due to the stresses created by the less than optimal conditions and a UV sterilizer won’t adversely affect plants like other treatment methods can. The second point worth mentioning, is the fact that Ultraviolet sterilizers also kill phytoplankton, the stuff that turns your water green.
Important Point: We have intentionally avoided the topic of bacterial infections in tilapia because these are not common in clean systems. However, it's worth mentioning that a UV sterilizer will also kill most harmful bacteria suspended in the water.
So there you have it. The answer to the question of what constitutes clean water and what can be done to keep it that way. But we're not quite finished with water yet. We still have to go over heating and filtration systems in general.
Filtration Components
Solids Separators: The most common type of barrier-less solids separator in aquaculture makes use of a phenomenon known as the "Tea Leaf Paradox". It was identified by Albert Einstein, so don't feel dumb if you've never heard of it or don't fully understand how it works. Basically, when you spin water in a bucket, the pressure of the water at the outside edge is greater than it is in the center. However, where the water touches the sides and bottom of the bucket, friction slows it down and the pressure drops. Since the water touching the sides and bottom can't keep pace with the rest of the water in the bucket, a boundary layer is formed. The water on the outside of the boundary layer takes a different path downward, towards even greater friction at the bottom. This secondary flow of water, aided by the pressure gradient of the spinning water, sweeps undissolved solids into a neat pile in the center of the bucket.
Separators that work on this principle are commonly referred to as swirl traps or swirl filters. In commercial aquaculture, these are normally constructed using cone-bottom tanks. On a smaller scale, these can be constructed from 25 gallon tubs. Another type of solids separator is known as a settling tank. There are several variations on this theme, but basically it's just a barrel through which water is passed and anything that is heavy enough, sinks to the bottom. The problem with settling tanks is that they are harder to clean and can only trap sinking solids. The final type of solids separator worth mentioning, is called a centrifugal separator. These separators work by spinning the heavier particles into a collection chamber where they can be flushed. These types of separators are only useful for removing the heaviest solids.
There is a good test that you can do, to determine what kind of separator you need. Simply fill a clear jar with the dirty water that you want to clean. Make sure to add some of the solids that you want to separate and put the lid on the jar. Shake the jar for a few seconds and then set it down, undisturbed and watch what the particles do. If all of your solids sink to the bottom within two minutes, you can use a centrifugal separator. If all of the solids sink to the bottom in five minutes or less, you can use a settling tank. However, if some of the particles sink and others float and some even hover in the middle, you will need to use a swirl filter. Spoiler alert... you're going to want a swirl filter.
Important point: Settling tanks are a lot like un-flushed toilets. The poop just sits at the bottom and dissolves into the water contributing to unsanitary conditions. Once tilapia poop has dissolved into the water, it is much more difficult to remove. When you also consider that freshwater fish like tilapia absorb water through their skin, it's no wonder that some backyard tilapia tastes like sh...
Pre-Filters: Also referred to in this guide as a coarse filter, is nothing more than a barrier that traps undissolved solids as water passes through. If you are using a swirl trap, then the pre-filter will serve as a secondary trap for solids that have a neutral buoyancy or otherwise escape. If you aren't using a swirl trap, then your pre-filter must be designed to handle a large amount of solid material. Drum filters are the most common types of pre-filters found on medium and large tilapia farms. Some drum filters are the size of city busses, while others are not much bigger than a recliner; it all depends on the amount of solid material being produced by the tilapia. Don’t be afraid to use your own ingenuity when it comes to pre-filters. There is nothing magical about commercially produced filtration systems. If you have the skills to make your own, by all means go for it. A good analog for a pre-filter is nothing more than a bucket, with holes drilled in the bottom, filled with polyester pillow stuffing, suspended over the pond and a pump to drop water through it. Of course, this isn’t very practical, because it would be time consuming to service and there are other filtration steps that need to happen, but the analogy is still accurate.
Aquaponics Point: You are running an aquaponics system to grow vegetables, not a fish sewage treatment plant. If you plan on eating your fish and you want them to be healthy and taste good, then do not allow any solid wastes to dissolve in your grow beds. Always use filtration to remove as much of the undissolved solid particles as you can, before they make it to your plants.
Note: I have taken a few critiques for the aquaponics point above. Purists will tell you to go ahead and allow the fish poop to enter the grow beds and use worms to compost the poop. They will tell you that mineralization of fish wastes will provide micro nutrients for your plants. My response is that I stand by my advice. The taste of tilapia raised in these veritable fish toilets is awful when compared to tilapia raised in clean aquaponic and aquaculture systems, free from decomposing fish poop.
Fine-particle filters: Use a bead filter, sand filter, diatomaceous earth filter or in-line water filter right after your pump, to trap dissolved solids. They are very effective at removing the particles that are too small to be trapped by any other filtration step. On a small scale, fine-particle filtration might not be necessary, due to the relatively low volume of water. This is especially true if you set up a pre-filter consisting of a swirl trap, followed by some compressed polyester pads. The biggest worry with this configuration will be an increased level of tannins and possibly phenols. In commercial tilapia farming, fine-particle filters are fitted between the water pump and the final water sterilization and polishing.
Biological filter: Not really a filter at all, its only purpose is to provide a large surface area on which nitrifying bacteria can grow. A box, with water in the bottom and some bio media, provides plenty of surface area for bacteria colonies to develop. There are a few design tricks to keeping a constant water level inside a plastic box, but it’s nothing that you can’t figure out if you decided to make your own. Biological filters should not need much servicing. In fact, you should avoid messing with them at all unless you notice that they are restricting the flow of water. This is why we prefer the wet/dry system, as opposed to the sand filters - the clear plastic makes it easy for us to see what is happening inside the bio filter.
Aquaponics Point: Flood and drain grow beds are the biological filter in aquaponics systems. In systems consisting of only floating rafts, a traditional biological filter will still be needed.
Important Point: As previously stated, There are certain tilapia farming situations where biological filters are not used. Do not use these alternative ammonia removal methods with aquaponics systems. Doing so will starve the Nitrosomonas and Nitrobacter thereby eliminating the supply of nitrates for the plants.
Ultraviolet sterilizer: This is not something that you should try to make yourself. Not because you might electrocute yourself, but because it probably won’t work. With UV sterilizers, the flow has to be just right. If it’s too fast, the parasites and algae will just fly right past the ultraviolet radiation and not even be affected by it, if the flow is too slow, it won’t kill them at a fast enough rate to keep up with their reproduction in your system. An ultraviolet sterilizer can also reduce the beneficial bacteria suspended in your water, so we don't recommend that you add a UV sterilizer until after your biological filter is established. Make sure to get one that is easy to clean. The clear tube separating the light from the water, also known as a quartz, needs cleaning from time to time. Some models come with a wiper system to do the job.
Be sure to understand that an ultraviolet sterilizer cannot cure any disease or remove any parasites or viruses that are already on or in, your tilapia. The only thing that a UV sterilizer does, is kill the organisms that are suspended in the water. It can be compared to putting a HEPA filter in a room with a sick person. It won’t do anything to cure the sick person, but it might help others from getting sick. That said; there is a difficult to understand benefit of ultraviolet sterilization known as "Redox" that happens at the molecular level and greatly contributes to the immune systems of tilapia and their ability to resist diseases. We promised to limit the science, so we'll let you look up "redox potential" on your own.
- A UV sterilizer can not cure sick tilapia; it can only reduce the spread of disease.
- A UV sterilizer will kill free-floating single cell algae in your system. This type of algae is not beneficial to fish or plants and can be dangerous due to its affect on carbon dioxide and pH levels.
- A UV sterilizer contributes to the "Redox" potential of your pond water, which greatly enhances the tilapias immune systems and their resistance to disease.
Water heating: Heating pond water during the cold months is the bane of every tilapia farmer. Factors such as incorrect species selection and improper pond construction, can force tilapia farmers to spend all of their profits or negate all of their savings, just to keep their tilapia alive in the winter. Death by cold water is the number one service call that we get in our area between January and March. If you’ve read elsewhere on our website, you already know how important selecting the right tilapia species is, but just as important, is proper pond construction. Tilapia ponds should be separated from the ground by some margin of insulation. Even if it's only an inch of foam, it's better than having the cold ground act as a heat sink for your pond water. Insulating the sides of your pond and covering the top with rigid foam at night, will help contribute to lowered heating costs. In colder climates or places where electric heat is not available or desired, a green house with a rocket stove may be the only solution.
When it comes to heating your pond water, you have two basic options: The direct heating method and the heat exchanger method. To use the direct heating method, simply put one or more heating elements into the water flow of your pond. Heating elements can be metal probe type, aquarium type or even simple water heater elements. Just use whichever one fits your system and budget, the best. With electric heaters, don’t get yourself hung up on individual wattage. Two 300 watt heaters do the same work and use the same electricity, as one 600 watt heater and so on. It might be more cost effective for you to buy several smaller heaters, instead of one big unit.
The second method of pond heating, is to use an external heat source and transfer that heat into the pond, using a heat exchanger. A heat exchanger can be made using a series of CPVC (hot water) pipes, running back and forth, covering the bottom of your pond or sump. Heated water is then pumped through the submerged pipes, warming the surrounding water. The source of heat can be a small water heater, more heating elements or even a solar water heater. Our favorite method to heat the exchanger, is to use a small water heater, with a circulation pump and a small pressure tank. A bright LED digital thermometer is also helpful. You might even install a low temperature thermal switch, to shut the circulation pump off, when the pond reaches a certain temperature. If you decide to try a solar water heating method, be sure to have an electric back up, just in case you get too many overcast days in a row.
So that’s it for clean water let's move on to the second thing that tilapia need.
Tilapia need number two - Oxygen
Critical Point: In this section, we will try to explain, in just a few paragraphs, what would normally take a college course to understand. Our original statement, that this is an answers-only guide, is especially true for this section. The conclusions that we present here, come from university studies and well-respected international research institutes. If you want more information, we highly suggest that you start your research at the Food and Agriculture Organization of the United Nations and then follow up with university research papers.
The air that you breathe is a mix of gasses, consisting of 20.95 percent oxygen (O2) and 78.09 percent nitrogen (N2). The remaining .93 percent is made up of other gasses (Ar, CO2, Ne, He, CH4, Kr, H2 and Xe). Most people know that water is made up of hydrogen and oxygen (H2O), so they assume that fish get their oxygen from the water molecules themselves. However, a fish's gills do not have the capability of separating the molecular bonds of water, so the oxygen in an H2O molecule is unavailable for respiration.
Surprisingly, the oxygen that fish breathe, is the exact same oxygen gas that you breathe. On land, your oxygen is delivered to your lungs "suspended" in an inert nitrogen gas; under the water, a fish's oxygen is delivered to their gills suspended in a hydrogen/oxygen liquid. It is mixed in with the water on a molecular scale. You would no sooner see the oxygen in the water, than you would the oxygen contained in the air that you breathe. This is called dissolved oxygen. Don't confuse dissolved oxygen with bubbles of any size, even the smallest bubble is millions of times larger than the oxygen molecules that fish use for respiration.
Since the oxygen, that is dissolved in water, is the exact same oxygen that is "dissolved" in the air, it would be logical to assume that oxygen can travel freely between air and water. Unless of course, we're talking about a calm body of water. Because you see, on a calm body of water, such as a pond, the water molecules near the surface act differently than the rest. Because they don't have any H2O molecules above them, to exert any attractive force, the top few layers of water molecules line up, pole to pole and form stronger bonds with each other. This force is known as the surface tension layer and it dramatically slows the transfer of oxygen entering and waste gases escaping, the water. An easy way to visualize the surface tension layer, is as a big sheet of plastic wrap, on top of the water, suffocating everything underneath.
On a moving body of water, such as a river, there is no surface tension layer. The constant churning of the water, continually drives the top molecules downward, breaking their bonds to each other. Without the surface tension layer, oxygen molecules can freely travel between the air and the water without any effort. Fortunately, for life in ponds, there are other forces that can drive the top layer of molecules apart, punching holes in the the surface tension layer and allowing for the free travel of oxygen and other gases. Strong wind or rain, for example, does a great job of breaking the surface tension. Also, bubbles, bursting at the surface, open holes in the top layer that allow for the exchange of gases.
The surface tension layer does more than just keep oxygen from entering the water freely, it also slows carbon dioxide and other gases from escaping. In tilapia ponds, carbon dioxide molecules are the by-product of fish respiration and organic decomposition. Carbon dioxide must be allowed to escape or the pond will stagnate and the oxygen-dependent life, will not thrive. Fortunately, the same actions that allow oxygen to enter the water, also allow carbon dioxide to escape. This is commonly referred to as the gas exchange. For tilapia farming operations, breaking the surface tension, to allow for an exchange of gases, is a requirement, not an option. It is the only way that carbon dioxide can escape freely and one of only two viable ways that oxygen can enter the water at an adequate rate.
As in all things tilapia farming, the method used to break the surface tension, known as surface aeration, is an economic decision. There are just as many ways to accomplish the task, as there are ways to share your money with retailers and manufacturers. Some methods allow for a high volume of gas exchange, but come at an unreasonably high purchase price and require a lot of energy to operate. Others are very cheap to operate, but do very little to facilitate an effective level of gas exchange. The effectiveness of any surface aeration method can be expressed as a ratio of the energy consumed to the surface area affected. The following methods, offer the best ratio of surface agitation to power consumption:
- Cascades, waterfalls and fountains are very cheap to operate and can be set up to break the surface tension over a very large area. To keep this method cost effective, do not restrict the water flow or lift the water very high above the surface. The water can be dropped down or jetted up, either direction is effective.
- Aggressive bubbling, that causes the water to be lifted, one inch or more above the surface. Use an air pump that can deliver more than 3 cubic feet of air per minute, at a minimum pressure of 6 pounds psi and a 2x2 inch coarse air stone. Do not waste energy trying to pump air through a fine air stone, such as a ceramic diffuser, this is the wrong application for fine bubbles. An efficient pump for this method will cost less than 2 dollars per month to operate.
- Paddle wheel aerators. For large ponds the paddle wheel aerator offers the lowest energy cost for the amount of surface area affected.
Critical Point: Don't get hooked in by marketing claims. Surface aeration is a multi-million dollar industry full of expertly crafted conjecture, that sounds reasonable to air-breathing humans. Any method to break the surface tension must be measured as a ratio of the energy used, to the surface area affected.
Nile tilapia need water with a dissolved oxygen content above three parts per million (ppm) and Blue tilapia need their oxygen above seven ppm. In a pond with a biomass of one pound for every 3.74 gallons of water, surface aeration will normally keep the dissolved oxygen level within a healthy range; even at 4:00 a.m. when the Diurnal change in dissolved oxygen concentrations are at their lowest. However, we recommend that a minimum oxygen density of 7 ppm (4 ppm for Nile) be measured once in the early morning (before sunrise) and then during the season at the warmest water temperatures. After it is confirmed that the dissolved oxygen content is above the minimum level at these times, a routine daily monitoring can be made, in the late afternoon. The daily late-afternoon monitoring will be different from the reading taken at other times and temperatures. However, as long as it is performed at the same time each day, it will provide a good benchmark to know when to take more sunrise or high-temperature readings; or to determine the need for supplemental oxygen.
Egghead Point: Sorry for dropping that Diurnal bomb on you in the paragraph above. It's just a fancy way of saying daily. But, we used the word diurnal to make the point that the science of dissolved oxygen is complex. Take the following formula for example:
O2¢ ¢ - O2¢ = P - R - Y ± A
where P = the oxygen produced via photosynthesis, R = the respiration of all living organisms in the pond including bacteria and plants, Y = the quantity of oxygen stuck in the sludge or mud at the pond bottom and A = the amount of oxygen dissolved from or released to, the atmosphere.
It's actually a simple formula for expressing the changes of dissolved oxygen over a period of time, expressed as t¢ ¢ - t¢. There are however, no shortage of very long and complex formulas for expressing the physics of oxygen in water.
So far we have limited the discussion to surface aeration methods. This is because surface aeration is all that is needed in recirculating aquaculture systems, with a biomass of 2 pounds per cubic foot, which can also be expressed as one pound per 3.74 gallons. It should also be mentioned, that certain non-recirculating systems, such as tilapia farming operations that divert river water, may also use surface aeration, in the form of a series of waterfalls, before the water is utilized. If the tilapia farming is being done in nets, suspended at the surface of a large body of water, such as a lake or very wide river, no surface aeration is normally necessary. However, if the suspended nets are floating in smaller bodies of water, such as ponds, surface aeration is still recommended.
- Biomass affects oxygen content
Biomass is the total of all oxygen breathing organisms in your water, including fish and bacteria. When we recommend a stocking density of two pounds of tilapia for every cubic foot of water, we are taking into account the typical shape of a recirculating aquaculture tank as well as the normal levels of bacteria. This density will allow the oxygen from the atmosphere to enter the water at a rate that will replace what is being used by the fish. Shallower ponds with a greater surface area exposed to the atmosphere can accommodate slightly higher stocking densities, while neglected, bacteria-laden ponds will only support lower stocking densities.
- Water temperature influences oxygen content
To illustrate how the water temperature can affect the amount of oxygen that the water contains, here's a practical comparison: At one standard atmosphere (760 torr), the oxygen saturation concentration at 35.6º fahrenheit is 13.86 ppm. Next, raise the water temperature to 60º fahrenheit and measure the dissolved oxygen again. It's dropped to 9.82 ppm. Finally, raise the same water to a temperature of 86º fahrenheit and the oxygen concentration drops to 7.44 ppm. As the water gets warmer, the amount of dissolved oxygen goes down.
- Light influences oxygen content
All bodies of water, including properly illuminated indoor tilapia ponds, have phytoplankton. They are tiny green algae that live suspended near the water surface. When the water is illuminated, the phytoplankton begin their photosynthesis, which in turn, gives off oxygen. This oxygen is easily dissolved into the water and by late afternoon, can significantly increase the amount of oxygen available to the tilapia. However, this condition is only temporary and as soon as the sun goes down or the lights are turned off, the phytoplankton stop producing oxygen. The result can be an oxygen drop to levels that are deadly for tilapia. This is why it is very important to measure the dissolved oxygen content at least once at 4:00 a.m., then later that afternoon at around 2:00 p.m. The morning reading must be above 7 ppm (3 ppm for Niles). Then, the afternoon reading can be used as guide to determine when to take another early morning reading.
- Decomposition depletes oxygen
The decomposition of organic matter uses oxygen and gives off carbon dioxide. This creates the worst possible scenario for tilapia farming. Without immediate intervention, this can wipe out an entire harvest in one night. The night time drop in the oxygen created by photosynthesis, combined with the continued oxygen consumption of decomposing organic material and subsequent release of carbon dioxide, which occurs around the clock, can cause the dissolved oxygen level to drop to almost nothing. This is another reason why it is so important to remove tilapia poop and uneaten food from recirculating aquaculture systems as quickly as possible, as part of the continual flow of filtration.
- Maximize surface aeration before considering alternatives
More often than not, low dissolved oxygen levels are the result of inadequate surface aeration. It's easy to forget that the gas exchange only occurs at the surface and only in the area affected by the aeration technique. For example, the spray from a fountain head only affects the area where the drops actually hit the water. So, if you have a pond with a surface area of 1800 square feet and you only aerate a six foot circle, you still have 1774 more square feet to work with. Tilapia don't care if you make it rain 24/7 on the entire water surface, they'd much rather breathe. Finally, when you've exhausted every surface aeration option and removed as much decomposing organic matter as you can, it might be time to consider thinning the number of tilapia in your pond.
Adding supplemental oxygen
Adding supplemental oxygen requires an oxygen source and a method to dissolve the oxygen into the water. There are only three oxygen sources to choose from and as you probably suspected, each has its own advantages and disadvantages. Bottled oxygen gas is the simplest to deploy and is the cheapest source of short-term oxygen. Make sure that it's medical grade oxygen, not oxygen intended for welding. Liquid oxygen is cheaper in bulk than oxygen gas, but it is a fire hazard, requires special training to handle and may require special permits to be on your property. In addition, liquid oxygen requires special equipment to make it suitable for use. Generated oxygen has the highest up-front costs, but over time, can save money over the other two oxygen sources. Generally speaking, the end result from every oxygen source is a tube, with oxygen gas flowing, under regulated pressure. It's pretty easy to understand.
The method used to dissolve oxygen into the water, on the other hand, is widely misunderstood. This, once again, stems from the fact that manufacturers are keenly aware that their customers don't understand the physics behind dissolving oxygen into water. The truth is, all that it takes to dissolve oxygen into water, is a hole in the ground, a couple pieces of pipe and some fittings. But, what manufacturer is going to tell you that their system, costing thousands of dollars, can be usurped with parts from home center store? Not to mention the fact, that the custom-built unit, is 100 percent efficient, wastes no oxygen, has infinite range of adjustment and can create dissolved O2 levels as high as 150 parts per million. Manufacturers would much rather capitalize on pseudo-science, selling snake oil remedies and fancy packaging.
Methods, such as flat-plate ceramic air diffusers, make very tiny bubbles. And, to the layman, make perfect sense. Supposedly, as the air bubbles slowly rise to the surface, the oxygen contained in each tiny bubble, comes in contact with the water and some of that oxygen is "dissolved". Okay, but assuming that the bubbles are full of pure oxygen, why don't they completely disappear? The truth is, most of the oxygen simply rises to the surface, where each bubble breaks a tiny hole in the surface tension layer and releases its oxygen into the atmosphere. Sure, a little bit of the oxygen gets into the water along the bubble's journey and it's certainly handy to have all that oxygen concentrated right there on the surface, when the tension is broken and the gas exchange occurs; but this method is not much more effective than surface aeration.
The other predominant method of dissolving oxygen into water is with the use of an oxygen cone. An oxygen cone works by bubbling oxygen up through a rapidly decelerating column of water. The bubbles of oxygen are held in place by the opposing forces (buoyancy vs. velocity), until they are absorbed. There are other variations on the oxygen cone theme, but this method is really the only other one that works, without wasting a lot of oxygen. The downside of oxygen cones, are their price and limited range of adjustment. For example, if the water flow is too strong, the bubbles get pushed out before they can dissolve; and if the water flow is too low, the bubbles will rise to the top, where they aren't effective.
U-Tube Oxygen Generation
The best method for dissolving oxygen into water is with the use of a U-Tube. This method uses hydrostatic pressure to effortlessly move a column of water through a gradient of increasing pressures that crush the oxygen into the water. This is nothing new, in fact, it dates all the way back to 1647, when Blaise Pascal first formulated the concept of pressure and how it is transmitted by fluids, such as water. The reason that you've never heard of this, is that there is no money to be made telling people how to do things for free; also, there's the fact that any search for u tube, ultimately takes you to a video sharing site.
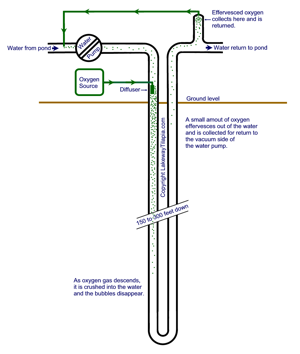
The construction of a u-tube is fairly straightforward. All you do is drill a hole in the ground anywhere from 150 to 300 feet down. You will most likely hire a well driller for this job. Since you aren't going to be drawing water out of the hole, you probably won't even need any permits, but check with your local officials just to be sure. After you have dug your hole run a long "U" shaped section of pipe to carry water down to the bottom and back up again. Thanks to the equalizing pressure on each side of the u-tube, a low horsepower pump is all that is necessary to push the water along. Remember, it's all about low energy consumption. Just make sure that the flow is fast enough to carry the bubbles down.
Click on the picture for a larger version, it's pretty self-explanatory. It can also be designed as a tube-within-a-tube, where the water travels down a smaller diameter center tube, but the plumbing connections on top will be more complex.
Tilapia need number three - Food
The fact that tilapia need food may seem just a little too obvious, for a guide that assumes its readers have an average level of common sense, but the amount of misinformation about feeding tilapia is appalling at best and deadly at the worst. Contrary to Internet lore, tilapia do not seek out poop as a food source. Tilapia farming operations in China have been observed feeding pig manure to their fish and the fish seem to eat it willingly. But what animal on earth won’t eat anything that appears to be edible, when it is offered no other choice. The truth is, just about all omnivorous fish will eat each others poop, as part of their inherent grazing and strike reactions. They aren’t swimming around the pond thinking "I could really go for some poop right now". The poop from pigs and humans is just plain disgusting. Like pigs, humans seem willing to eat just about anything, including the poop of many sea creatures, including oysters, clams and shrimp. Don’t even get me started on the humans who drink the water squeezed from elephant crap or eat dung beetles. It’s no wonder that we seem willing to believe that a tilapia would consider poop a savory edible, considering all the fecal material that we pay good money to eat in our lifetimes.
Critical Point: Don't confuse the above statement about Chinese fish farms with the practice of "fertilizing" algae growth in ponds, as the Chinese would have you believe. There is a big difference between suspending chicken coops over ponds to promote algae growth and what the Chinese fish farmers are doing. Incidentally, fertilizing ponds with manure is still practiced today, in spite of a very exhaustive Taiwanese study proving it ineffective.
So what do tilapia eat? Well, tilapia are omnivores, but they have very strong tendencies towards being vegetarian. The tooth and jaw structure of a tilapia is designed to graze on algae and other aquatic plants. If you want to observe accelerated growth in tilapia fry, put them in an algae-covered aquarium, next to a sunlit window. They will devour the algae, growing much faster than fry that are only given a commercial omnivorous fish food. Here at our hatchery, we feed our newly hatched fry spirulina algae discs to get them up to size quickly. This also gets them out of the "danger zone" faster, since tiny fry are far more delicate than larger fingerlings.
Just about everyone knows that tilapia need food to grow and it's not much of a stretch to understand that the more your tilapia eat, the faster they will grow. Although technically, just eating the food isn’t the secret to growth; it needs to be metabolized with the aid of oxygen, proper water chemistry and temperature, as stated earlier in this guide. However, for the purposes of our explanation, we’ll just say that more food equals faster growth. One thing that catches new tilapia farmers by surprise, is the practice of using less food to slow growth. The main reason for slowing growth, especially in large juveniles, is to hit a target harvest date. It should also be noted, that this practice should be carefully administered for just a couple of weeks to avoid the risk of permanently stunting the growth of the tilapia.
Nothing contributes to tilapia health more than good nutrition. The proper diet will boost their immune system and help them resist disease. When combined with an ultraviolet sterilizer, to boost the Redox potential in your pond, proper nutrition will make your tilapia ready for just about anything. But, what constitutes proper nutrition? Well, when you consider that thousands of years of evolution have adapted their physiology to get everything that they need from algae and aquatic plants, then aquatic greens are the answer. Unfortunately, tilapia eat algae and plants much faster than they can grow back in a small area. In the wild, tilapia schools graze over several miles. A commercial tilapia farmer, intent on feeding only aquatic greens, would need to dedicate several square feet of water surface area, to grow sufficient food for a single tilapia. As with all commercial livestock farming, dedicating acres of valuable land to serve as the sole source of animal food just isn’t practical and just about every farmer supplements or completely replaces, the livestock’s natural diet with a nutrient-dense manufactured food.
While not exactly what evolution has designed them to eat, tilapia do extremely well on some commercially produced food. The consistency of a manufactured diet offers many advantages to the tilapia farmer that a natural diet would not. The even distribution of nutrients and uniformity of size, goes a long way to ensure that every tilapia in the pond gets the same level of nutrition. The amount of food to give is determined by the weight of the fish and the temperature of the water. Uniformity between individual bags of food, keeps projected growth rates and harvest dates, on track. Best of all, some manufactured tilapia food is scientifically designed for the fastest growth possible, when a proper feeding schedule is followed. So now the question is, how much food do tilapia need?
To determine how much food to feed tilapia, you need to know three things: The water temperature, the average weight of each tilapia and the the biomass; which is just a fancy word for the total weight of the living organisms per cubic foot of water or for our purposes, just the total weight of all of the tilapia. As the water gets colder, tilapia metabolize food slower and grow slower so they need less food. The opposite is also true as the water gets warmer. During the early stages of growth, up to about 2 ounces, tilapia are little eating machines that can devour a much higher percentages of their body weight per day. But as they grow, that percentage goes down. Obviously, since you don’t feed tilapia individually, it’s helpful to know the total weight of all of the tilapia in your pond, so that everyone gets to eat their fill.
There are a lot of scientific calculations that you can do to determine the perfect amount of food to give each day and if you're inclined to do all of the math yourself, we urge you to continue on your quest to become the ultimate tilapia nerd. For the rest of us, there are charts and graphs, made by other nerds. Here’s one for Purina AquaMax, the most nutritionally advanced tilapia food in the world.
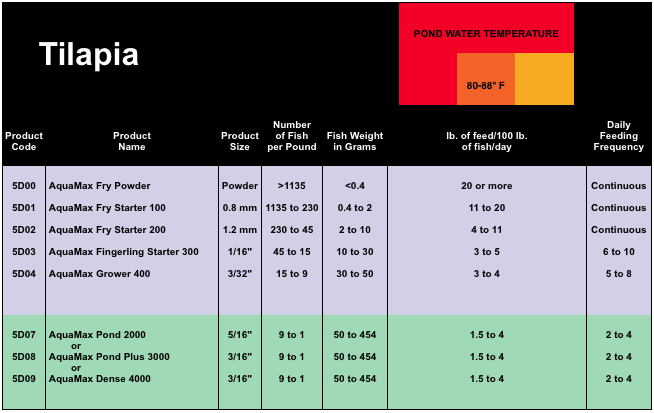
The Purina chart is a bit generalized, but it’s still a decent guide for using their products and it illustrates a couple of important facts about tilapia food consumption in general. Compare the Fish Weight in Grams column, to the Product Size column and you’ll see that as the weight of each individual fish increases, the size of the food grains also increases. This part is obviously because bigger mouths can eat bigger food. Now, have a look at the column titled lb. of feed/100 lb of fish/day. That’s just another way of saying "percentage of body weight to feed". All of the numbers in that column can also be read as a percent. For example, 20 percent or more, 11 to 20 percent, 4 to 11 percent and so on. Notice that, as the tilapia get bigger, it takes fewer individuals to make a pound of fish and the percentage of food to body weight goes down. This is because as tilapia get bigger, their rate of growth slows. Finally, notice the red area on the chart that shows the optimal feeding water temperature of 80-88 degrees Fahrenheit. As tilapia get colder, they metabolize less food and therefore eat less. Another reason why selecting the right tilapia species for your operation and giving some thought to your harvest dates, is so important.
Warning: Like everything else in the tilapia farming world, there are opportunists trying to turn a quick buck selling marginal nutrition as premium fish food. Most of this food is custom labeled, mass-produced, generic garbage composed of farming wastes. Anyone who wants to start their very own fish food company, can have their name and logo put on the bag. There's even an organic version that contains a plethora of indigestible ingredients, including peat, clay, diatomaceous earth, granite dust and lots of metal oxides and sulfates. Whatever you save using their low cost food today, you will lose as a result of an extended grow-out period. It's definitely not a good choice. We urge you to check out our Tilapia Feeding Guide to learn more.
There is an alternative method that is more complicated to calculate but far more accurate than any food manufacturers feeding chart. However using this method requires that you know the weight of your tilapia in grams. There are a couple of methods for weighing fish, but for feeding purposes weighing a random sample and then extrapolating that weight into the total number of fish will get you close enough. So if you know that you have 200 fish and you can weigh out ten of them, you can multiply the weight of those ten by 20 to get the weight for all 200. Of course, if the tilapia are small enough to all be weighed at the same time, then that would be the most accurate. By the way, the best method for weighing tilapia of any size, is to first weigh out a bucket of water to a known weight using a digital scale, then add the tilapia to the water and weigh again. The increase is the weight of the tilapia.
The next step is to determine the normal growth rate. If your tilapia are between two and five inches long, the average growth rate is 4% per day. If your tilapia are between five and six inches long, the average growth rate is 3% per day. If your tilapia are over six inches, and less than eight months old, the average growth rate is 1.5% per day. If your tilapia are between eight and twelve months old, their growth rate is .5% per day. If your tilapia are older than one year, feed them whatever they will eat in five minutes twice per day as their growth rate can no longer be measured in days.
Now that you know their weight and growth rate, you want to give them their weight in food times their percentage of growth for the day. So if you have 1,000 grams of tilapia that are all three inches long, you will want to start off by feeding them with 40 grams of food (1000 x .04). If you are starting with 1,000 grams of 5-inch tilapia, you'll want to start off by feeding them 30 grams of food (1000 x .03). If you have 1,000 grams of tilapia that are seven inches long and under 8 months old, you can start them with 15 grams of food. Now comes the tricky part, because on the next day your fish will weigh a little more. How much more? Well believe it or not, they should be the previous days weight plus their growth rate. So not only did you give your three-inch tilapia fingerlings 40 grams of food, they grew by the same amount and the next day you can calculate their new feeding values using their new weight of 1,040 grams.
Remember the golden rule of feeding: that if your tilapia can't eat all of their food in under 5 minutes, feed them less. A couple of factors that can affect how much food your tilapia will eat are temperature and disease. Look for signs that you are over feeding, such as uneaten food or filters becoming abnormally "full" in a short period of time. If you lower the amount of food being given and there is still uneaten food, take a careful look at your tilapia for signs of disease; such as swimming slowly or lethargy, an apparent lack of fear of your hand, lack of buoyancy, sores, etc. Check their water temperature to make sure that it’s not near the limits of their survivability range. If everything checks out, then reduce their food even further. Remember that tilapia can go for several days without food, so don’t be squeamish about lowering their food until it’s all being eaten. Oh and please re-check your calculations. Many tilapia farmers have accidentally forgotten the zero and multiplied the weight by .4 instead of .04.
We sell three varieties of Purina AquaMax in addition to spirulina algae discs on our Tilapia Food For Sale page. And if you're looking for some professional day-to-day feeding charts, have a look at our Tilapia Feeding Guide.
Tilapia need number four - Light
If you’ve ever seen an aquarium of tilapia fingerlings at night, the sight is rather disturbing. Hundreds of fish swirling around, like dead bodies, seemingly trapped in the invisible underwater currents. When you first turn on the lights, the only way that you’ll know that they aren’t all dead is that they are upright, instead of upside down and sideways. It’s very clear that tilapia need light to survive. Without light, they won’t move or eat and they will die. So the question is, how much light is needed?
In aquariums, tilapia can be observed hovering in the path of a beam of sunlight, as it shines through their water. In aquaculture ponds where there is a mix of direct sun and shade, tilapia seem to prefer the sunny side over the shaded side. There are several explanations for this behavior; many of them plausible. But whichever theory you are inclined to believe, it's obvious that tilapia prefer a bright, pond-filling, light.
At our hatchery, we provide our tilapia with 18 hours of light per day, using a combination of sunlight and electric light, that stays on until midnight. Why? Because the longer that tilapia have light, the longer they will stay active; the more they will eat and the faster they will grow. There are a lot of tricks to running a successful hatchery (or farm) and using light to extend the hours of food metabolism is one of them.
Of course, the best light that you can give to your tilapia comes directly from the sun. In addition to being a very powerful source of light, sunlight can be directed with the use of solar tubes and mirrors, to create pond-filling illumination. In outdoor ponds, brightly illuminated shade is just about right. The kind of light found inside a plastic covered cold frame greenhouse, is another great example. If you can provide partial direct sunlight for your tilapia, that's even better. On top of everything, sunlight is completely free, automatically making it the best choice for commercial tilapia farming. In fact, the only downside to sunlight, is the unwanted wavelengths of light that come with it, such as Ultra Violet and Infrared.
The second best lighting source for any pond, commercial or residential, is one that delivers photosynthetically active radiation or "PAR". These are the lights used by hydroponic and aquaponic growers, because they deliver the full spectrum of light used by plants for photosyntheses. They do not emit the photons (light) that can be damaging to cells and tissues, like shorter wavelength lights can; and for the most part, the entire PAR spectrum is within the visible range of the human eye. In other words, they're pretty safe for humans and fish. These are also the preferred lights to use for "extending the day" for fish activity. In addition, they work perfectly to grow plants, if that is part of your tilapia farming operation.
PAR lighting comes in many different forms. Some of the most popular are High Intensity Discharge (HID) types, such as High Pressure Sodium (HPS) and Metal Halide (MH) For commercial tilapia farming, HID lights are preferable, due to their intensity, which allows the light source to be placed farther from the water. Other options, such as PAR spectrum fluorescent lights are inexpensive. However, their relatively low output, requires that they be placed closer to the water surface than HID lighting. Newer technologies, such as LED and Plasma, use much less energy and produce very little heat. Unfortunately, they also come with a very high price tag.
As a last resort, you can use single wavelength fluorescent lighting, provided that they are daylight balanced to between 5,000 and 5,500 degrees Kelvin. In case you didn't already know, Kelvin is a color temperature, not a measure of heat or wavelength, as previously mentioned. It's comparable to the hue of a light source, if that helps you understand it better. Sunlight has a color temperature of between 5,000 and 5,400 degrees Kelvin and overcast skies are 5,500 to 6,000 degrees Kelvin. You can get daylight balanced fluorescent bulbs at any home center store; you do not need to buy expensive aquarium lighting. Just as important as the color temperature is the actual wattage. Your bulbs need to have enough power to cut through the water and light the bottom of your pond. Even still, fluorescent lighting pales in comparison to direct or indirect sunlight and HID lighting.
Tilapia need number five - Room to swim
Tilapia tolerate crowded conditions better than most species of fish, but they do have their limits. Increased numbers of tilapia can easily deplete the shared oxygen supply faster than it is being replaced. Oxygen that hovers at barely survivable minimums can cause damage to organs and other sensitive tissues, leading to illness. Overcrowding causes stress that leads to slower immune system response and poor resistance to disease. In addition, lowered oxygen levels also reduce the Redox potential of water, making tilapia even more susceptible to pathogens. The triple whammy of stress, reduced oxygen and lowered Redox, are an open invitation for diseases like Streptococcus, Aeromonas or Columnaris, none of which can be cured economically.
In a clean water pond, normal surface aeration will support a density of two pounds of tilapia for every cubic foot of water. That's a one pound tilapia for every 3.74 gallons of water. With the use of supplemental oxygen, a density of five pounds per cubic foot can be achieved. The highest documented tilapia farming density that we have found, was seven pounds per cubic foot. However, this was an experimental system, that utilized liquid oxygen to raise the O2 levels above 150 ppm.
Reality Point: It is being falsely stated, by several tilapia fingerling sellers and aquaponic systems dealers, that a "density" of one fish per gallon of water is "what everybody does". This is a marketing fabrication and is only possible if they intend for their customers to harvest their tilapia when they reach ¼ of a pound, yielding a couple of one ounce nuggets.
It's important to distinguish between the volume of water in a system and the area of water available to the tilapia. While the volume of water plays a role in the available dissolved oxygen, it does not have an affect on the stresses caused by the close quarters of an overcrowded environment. Even in open water tilapia farming, where the tilapia are raised in suspended nets, with potentially endless dissolved oxygen, over crowding can lead to disease, food suppression and slowed growth. To combat food suppression in crowded ponds, Purina makes an AquaMax food specifically for dense tilapia farming. The smaller pellet size means more pellets per pound of food and gives every tilapia a chance to grab a mouthful at every feeding.
Critical Point: Some small tilapia farmers use volumizing tanks or have a large amount of water contained in aquaponic floating rafts, but this water should not be considered when calculating the density of a tilapia pond. Only the area that is occupied by tilapia counts.
So that's it for the five needs of tilapia, let's move on to part 2 - tilapia farming systems.
